By Dr. Ted Lampidis
Most medical researchers never live long enough to realize that what they have accomplished has contributed to humanity in the form of a life-saving procedure, medical device, drug, or vaccine. Perhaps the best example of this is the Salk vaccine for the prevention of the catastrophic disease known as polio, which in the 1950s infected hundreds of thousands and either killed, permanently crippled or left millions fighting for their lives lying in iron lung machines.
Unfortunately, many of those scientists that first worked on uncovering the intricacies of how the immune system works or how a virus is able to infect a host cell – and all the pathways required to be activated and employed to produce more virus necessary for the Salk vaccine to be created – never lived long enough to see how their efforts ended up saving millions of lives.
So, I consider myself very fortunate to see that the sugar analog, 2-deoxy-D-glucose (2-DG for short) that I have been researching and developing for human use for the past 40 years, from my days as a post-Doc researcher at the Dana Farber Cancer Institute, is being used today to treat stage 4 cancer patients in a number of countries around the world.
2-DG is unique in that by mimicking glucose, it preferentially accumulates in cancer cells, and once inside the cell, it blocks the further use of glucose thereby shutting off a vital energy source as well as the material required for a cell to multiply. Similarly, it lowers the energy and shuts down the building blocks required for a viral-infected cell to produce more virus.
When I started my research, I had no idea that eventually this sugar analog known as 2-DG would be approved by the Indian government to be used to treat COVID patients. Although in retrospect, there have been several reports as early as the 1950s indicating increased glucose metabolism is a common trait among a wide variety of cells infected with different viral types.
The fact that 2-DG preferentially accumulates in cancer and viral-infected cells by exploiting a natural trait, common to both of these different cell types, sets it apart from other anti-cancer and anti-viral drugs that enter equally into cancer, normal, viral-infected and non-infected cells.
Thus, there is growing awareness of how unique and powerful a drug 2-DG is, and the increasing number of medical applications it has. In this regard, in a recent review written in collaboration with my dear departed colleague Dr. Enrique Mesri at the University of Miami, the science upon which 2-DG came to be used to treat COVID and other viral diseases is described.
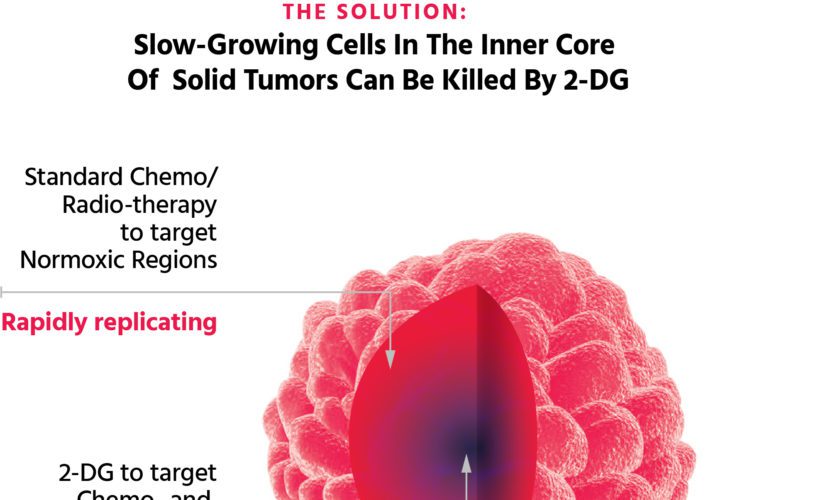
In this review, we detail how our surprising findings that 2-DG kills select tumor types even when growing under normal oxygen levels, have been applied to inhibiting KSHV, the virus responsible for inducing Kapos’si Sarcoma the most prevalent cancer in AIDS patients. The mechanism uncovered was that 2-DG acting as a mannose analog interferes with a process known as n-linked glycosylation. This process involves the attachment of a string of sugars (oligosaccharides) to proteins to make a glycoproteins which then attach to the outside of a cell where they perform different functions.
When 2-DG interferes with this process, it induces a form of stress – referred to as “ER stress” – that leads to a shutting down of viral replication. This is in addition to the aforementioned action of 2-DG acting as a glucose analog and inhibiting the building blocks required for viral production. Thus, it appears that 2-DG has more than one way to block viral replication.
With regard to 2-DG interfering with glycosylation, it was recently announced that the Noble Prize in chemistry was awarded to Dr. Carolyn Beertozzi for her work in glycobiology where one of her breakthroughs was on the interference of glycosylation of cell surface glycoproteins to enhance immunotherapy.
This follows the recent publication in an issue of Science Translational Medicine, where it was shown that 2-DG was effective in enhancing CAR T-cell therapy by breaking down the glycosylation shield that surrounds tumors inhibiting the access of immunotherapy. Several of our lab’s publications were cited in this article for the role we played in uncovering 2-DG’s effects on glycosylation as it applies to cancer.
This recent data further strengthens the significance of our work on 2-DG, demonstrating that in addition to mimicking glucose and inhibiting glycolysis, as an analog of mannose, it interferes with glycosylation which can be used to enhance cancer treatment as well as potentially treat a number of viral diseases such as COVID, herpes and others.
Below are excerpts from a recent review (2021) on glycobiology and glycosylation highlighting the importance of the emerging new field of glyco-immunotherapy. “Cancer immunotherapy has revolutionized treatment and led to an unprecedented wave of immuno‐oncology research during the past two decades. …However, the challenge in the coming decade is to develop cancer immunotherapies that can more consistently treat various patients and cancer types…The role of aberrant glycosylation in this process, and how it influences tumor immunity and immunotherapy is beginning to emerge… We discuss these insights in the context of clinical findings and provide an outlook on modulating the regulation of glycosylation to offer new therapeutic opportunities. Finally, in the coming age of systems glycobiology, we highlight how emerging technologies in systems glycobiology are enabling deeper insights into cancer immuno‐oncology, helping identify novel drug targets and key biomarkers of cancer, and facilitating the rational design of glyco‐immunotherapies. These hold great promise clinically in the immuno‐oncology field.”
Based on the recent events described above, it appears that the science behind 2-DG justifies the title of a review that my students and I wrote several years ago – The Wonders of 2-Deoxy-D-glucose – which highlights some of the salient features of this truly remarkable sugar analog. From that review, the following excerpt summarizes our thoughts on why 2-DG is such an important molecule: “Through the eons of time, out of all possible combinations and configurations of hydrogen, carbon and oxygen, nature has selected glucose not only as a vital source of energy to sustain life but also as the molecule who’s structure supplies the appropriate elements required for a cell to grow and multiply. This understanding, at least in part, explains the profound effects that the analog of glucose, 2-deoxy-D-glucose, has been shown to have on as common and widespread diseases as cancer, viral infection, aging-related morbidity, epilepsy and others.”






 Dr. Theodore Lampidis Laboratory located at Pap 124 receiving the award.
Dr. Theodore Lampidis Laboratory located at Pap 124 receiving the award.